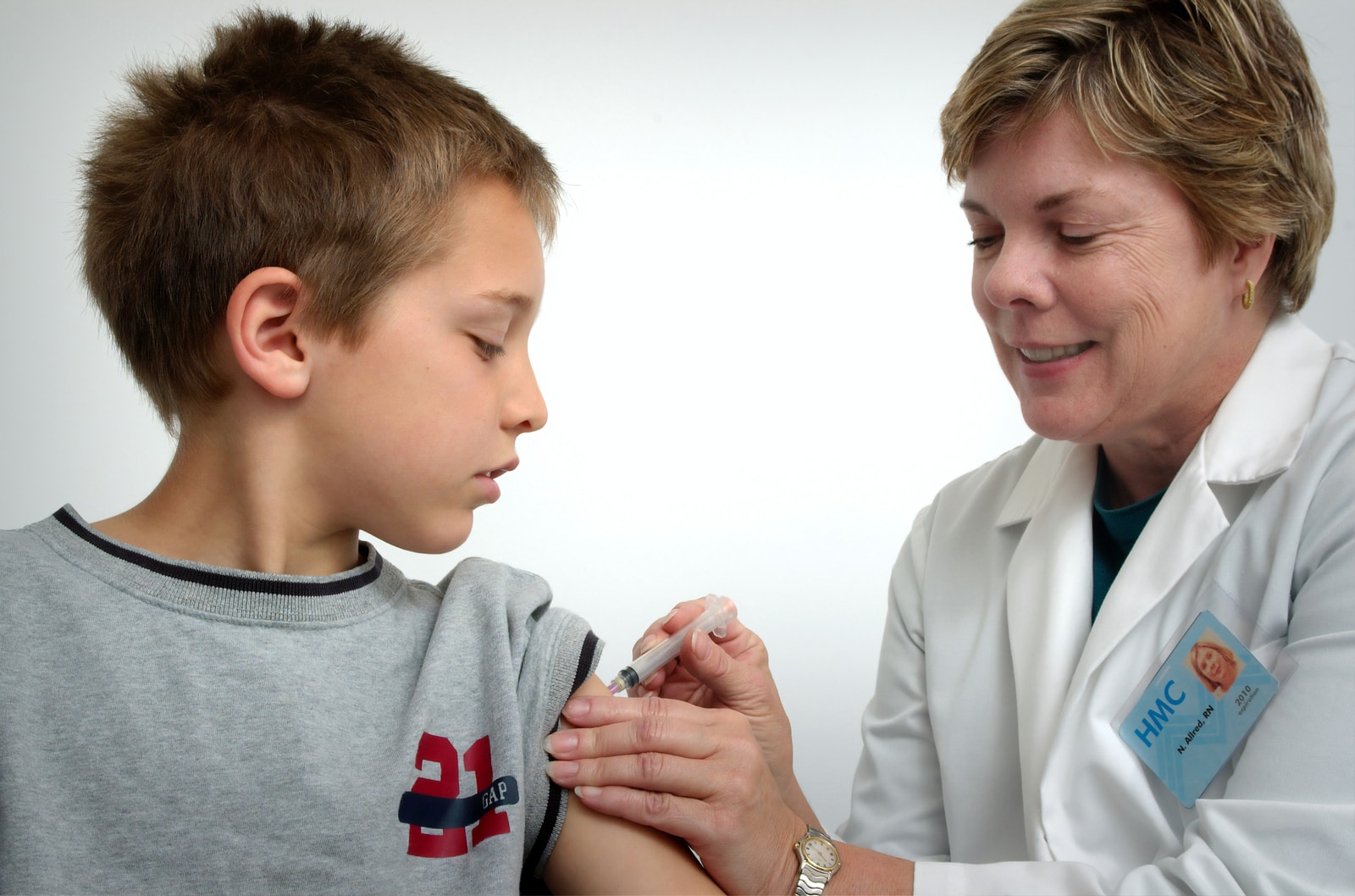
There is currently no COVID-19 vaccine approved for use in children younger than 12. This is a concern for many parents, who are sending their kids back to school in the face of the rapid spread of the highly contagious delta variant. These parents were able to see a glimmer in the future when the full approval by the Food and Drug Administration of the Pfizer BioNTech vaccine was granted on August 23. This is because vaccines and drugs that have been FDA approved can be used off-label. Doctors could give lifesaving shots to anyone outside the approved population. Many hoped this would mean that doctors would start giving the two-dose COVID-19 vaccine, even to children younger than 12.
Although it is legal to give the shot off label, the Centers for Disease Control and Prevention (FDA), and the American Academy of Pediatrics (AAOP) recommend against the vaccination of children under 11 until more data is available about how safe and effective the vaccine can be used. They also reprimand doctors who administer the vaccine off-label.
In a press conference, Janet Woodcock (FDA commissioner), stated that “we don’t have data on how much to give, nor the safety of children younger than the EUA.” We are not suggesting that children under 12 years old be vaccinated. It would not be appropriate. She said, “They’re not small adults.”
FDA approved the Pfizer vaccine’nbsp. The FDA approved the vaccine, now known as Comirnaty, for adults 16 years and older. It is not yet approved for children 12-15 years old. This age group can still receive Pfizer shots because the vaccine was approved for emergency use by 12 years old and above. The CDC recommends that everyone aged 12 and over get vaccinated against COVID-19. Children under 12 years old are at greatest risk from off-label usage.
Shortly after the news of the FDA’s full approval broke, there were reports of parents of children under the age of 12 calling their pediatricians and even using Twitter to ask if their kids could receive the shot off-label. One parent of triplets aged 11 years old told The Wall Street Journal that she would get her children an off-label vaccine.
Hannah Lichtsinn MD is a Minneapolis-based pediatrician and intern medicine practitioner. She says she can understand the feelings of parents. Dr. Lichtsinn states, “As a parent with young children, I want my kids to be safe from injury, sickness, and harm. Also, I really want them to be in school and continue their education this year because they missed so much.” “That desire is what drives parents and their doctors into exploring every way to keep children healthy during this pandemic,” Dr. Lichtsinn says.
Currently, trials into vaccine safety and efficacy in children are underway. Safety data . is expected to be available in September .2021. Many experts, including Dr. Lichtsinn believe that vaccine safety data for children ages 5-11 will be available in September. “The phase one trials in children looked at a range of doses to determine which was most effective, so the dose in younger children will no doubt differ from those in older children,” said Paul A. Offit MD, director of Vaccine Education Center, and a professor in pediatrics at the Children’s Hospital of Philadelphia.
“The American Academy of Pediatrics has cautioned against giving the vaccine off-label [to children under 12 years old] because they want to avoid, one) the flood of parents calling their pediatricians asking for it and, two), the burden that it will place on doctors to do dosing and schedules a la carte,” said Jessica Malaty Rivera, a Boston Children’s Hospital infectious disease epidemiologist. It could make it very difficult and potentially dangerous to not have it standardized.
“The good news? These trials are ongoing, very important priority and I am optimistic we won’t have to wait longer to have high quality evidence that says this product is safe in children,” states Paul Pottinger, MD, an infectious disease specialist at the University of Washington School of Medicine. It is disappointing that we are not there yet, especially since schools are reopening. However, that information will soon be available.”
There is precedent for off-label vaccinations. However, this is usually when doctors give a vaccine for someone under the age of 50. John D. Grabenstein, PhD is the general manager of Vaccine Dynamics and editor for Immunization Action Coalition. He is also an expert on the .COVID-19 Vaccine Analysis Team. Malaty Rivera said she’s heard from parents who were 11 years old and are interested in getting the vaccine off-label.
“I think it would be very reasonable to vaccinate someone who’s 11-and-a-half, or 11-and-three-quarters [years old],” Dr. Lichtsinn says. “There is no reason to believe that the vaccine will behave differently in a child this age than it would in someone who just celebrated their 12th birthday.”
Dr. Pottinger acknowledges that the age cutoffs can seem “arbitrary”, and in some ways they are. Children develop at their own pace. He stresses that “but this is how the studies were designed.” It’s biologically plausible and probable that an 11 year old would do well. However, that’s not how the studies were conducted. We have to respect science and the regulatory process. I am hopeful that we will soon have technology that can be used for 11-year olds.
Dr. Pottinger recommends that parents of children with compromised or other medical conditions talk to their doctor about their options. It’s unlikely that their doctors can do much. While off-label Pfizer vaccine use may be legal, providers who administer the vaccine off-label will be censured effectively by the CDC. “Clinicians who administer COVID-19 vaccines off-label .to children under 12 years would be violating their provider agreement, risking liability for adverse events, and potentially forfeiting payment,” the American Academy of Pediatrics noted. A doctor who prescribes off-label is leaving themselves at risk for being sued, and could lose their eligibility to be in the CDC’s COVID-19 vaccination program, meaning they may no longer be able to administer vaccinations to anyone, The Wall Street Journal reports Dr. Pottinger says that, although some parents may find the regulations and system frustrating, they are safe and work.
Malaty Rivera states that although she can see some flexibility for an 11-and-a half-year-old, she doesn’t recommend parents trying to convince their pediatrician to give an off-label shot. Malaty Rivera states that her children are aged 4 and 3 and she doesn’t feel comfortable asking for an off-label prescription. She also says that she would rather have my children enroll in a clinical study that is still enrolling, or wait for authorization. She adds that when the Pfizer vaccine is given an emergency use authorization, as experts expect it will, high-risk or .immunocompromised children .should be prioritized.
Although some parents may not wish to wait, September is only a few weeks away. The good news is that we will soon know when the trial data will be available. Dr. Pottinger states that parents should get immunized and make sure everyone in the school environment is immunized.